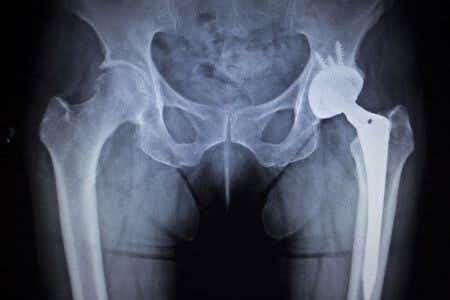
This case involves a 70 year-old woman with a history of rheumatoid arthritis who used the Zimmerman hip device after she fell at her home and dislocated her hip. After assessing her condition, the doctor decided a hip replacement was her best option. The operation went well and the patient was allowed to leave after four days. However, she soon started to experience pain and swelling in her hip area but did not inform her doctor until the pain became severe. A later evaluation showed she had developed metallosis since receiving the Zimmer hip replacement. She subsequently questioned the safety and regulations used for the Zimmer hip.
Question(s) For Expert Witness
1. Have you reviewed cases involving the Zimmer hip device? If yes, please explain.
2. Have you ever been involved in the approval process for this type of device or similar devices? If yes, please explain.
Expert Witness Response E-007633
I have a PhD in Biomedical Engineering and am a former FDA Lead Medical Device Reviewer. I used to be an instructor for Medical Device Regulation classes at a major state university. I have reviewed cases like this involving Zimmer, as well as similar issues with other manufacturers. I was a lead medical device reviewer at the FDA in Cardiovascular (predominantly Class III products, and some Class II). I also served as the FDA representative for the ASTM F04 task group, charged with the development of new testing standards for all medical devices.
About the author
Joseph O'Neill
Joe has extensive experience in online journalism and technical writing across a range of legal topics, including personal injury, meidcal malpractice, mass torts, consumer litigation, commercial litigation, and more. Joe spent close to six years working at Expert Institute, finishing up his role here as Director of Marketing. He has considerable knowledge across an array of legal topics pertaining to expert witnesses. Currently, Joe servces as Owner and Demand Generation Consultant at LightSail Consulting.