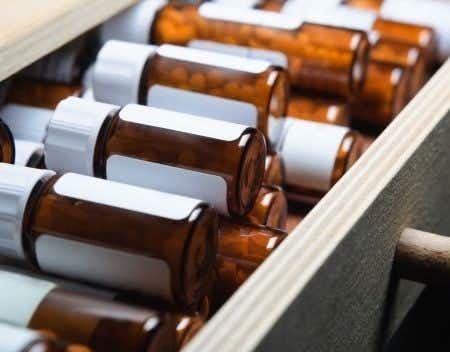
This is a case against distributors & manufacturers of opioids for, among other things, the violation of the Controlled Substances Act. Under the Controlled Substances Act, distributors are responsible for ensuring that opioids aren’t distributed and sold under suspicious circumstances, i.e. a closed loop system. The defendants are alleged to have not monitored the sale of drugs and did not report suspicious circumstances regarding their pharmacy customers. These distributors have been fined repeatedly for violating the CSA. We require an expert who is familiar with DEA diversion investigations to discuss the process and opine on how pharmacies attempt to avoid these investigations and continue to enable the opioid epidemic.
Question(s) For Expert Witness
1. Please briefly describe your experience with the DEA.
2. Can you speak specifically to diversion investigations? Do you have experience with opioids in this context?
Expert Witness Response E-007703
The DEA is required to establish and maintain a closed system of distribution for the Importation, Manufacturing, Distribution, and Dispensing of all controlled substances. The system is designed to provide accountability for all controlled substances from start to finish. The DEA is also responsible for placing quotas and restrictions on all products that enter the United States as raw materials for production, security measures imposed on distributors, and the dispensing to the end-user. The DEA, and more specifically, the Office of Diversion Control, enforces the Code of Federal Regulations that is consistent with the Controlled Substance Act of 1970. The DEA maintains that all entities who either purchase or prescribe controlled substances be registered with the DEA. Each registered entity must be in compliance with rules they must be followed in order to maintain an active registration. Registrants who don't comply face Administrative, Civil, and/or Criminal Sanctions. For example, Pharmacists have a corresponding responsibility and must only fill valid prescriptions for controlled substances issued by a legitimate practitioner for a legitimate medical purpose.
About the author
Joseph O'Neill
Joe has extensive experience in online journalism and technical writing across a range of legal topics, including personal injury, meidcal malpractice, mass torts, consumer litigation, commercial litigation, and more. Joe spent close to six years working at Expert Institute, finishing up his role here as Director of Marketing. He has considerable knowledge across an array of legal topics pertaining to expert witnesses. Currently, Joe servces as Owner and Demand Generation Consultant at LightSail Consulting.